Discoidin Domain Receptor (DDR) inhibitors
A Potential Novel Therapeutic Class for Fibrosis
Discoidin domain receptors (DDRs) were discovered in the 1990s but more recently they have gained traction as new therapeutic targets with potential to treat multiple fibrotic conditions.
DDRs are receptor tyrosine kinases containing a discoidin homology domain in their extracellular region, which bind to collagen. There are two DDR receptors, DDR1 and DDR2, which act as non-integrin collagen receptors.
On binding of collagen, the DDRs autophosphorylate, initiating various downstream signalling pathways that drive clustering, upregulation and further collagen synthesis. DDR expression is increased in many fibrotic diseases and preclinical proof-of-concept for small molecule inhibitors has been demonstrated in preclinical models of lung and kidney fibrosis.
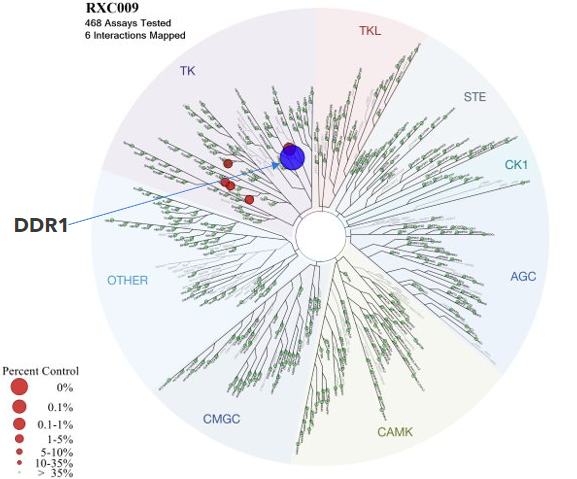
We have developed novel, orally available, selective and potent small molecule DDR inhibitors with drug-like properties, and in October 2023, nominated our DDR1 selective inhibitor as development candidate RXC009, which has the potential to be a first-in-class treatment option for chronic kidney disease.
Currently, there are no selective DDR1 inhibitors in clinical trials, although several kinase inhibitors developed for other kinases have shown varying degree of DDR specificity. We presented a poster on a novel, selective DDR1 inhibitor, REDX12271, at the American Society of Nephrology (ASN) Kidney Week, November 2022, and we returned to ASN in November 2023 to present RXC009 preclinical data from a therapeutic Unilateral Ureteral Obstruction (UUO) murine model of kidney fibrosis.
The data presented showed that RXC009 treatment resulted in:
- A significant reduction in histological markers of both inflammation, kidney injury and fibrosis.
- Target engagement in kidney tissue was also demonstrated in phospho-DDR1 (p-DDR1)
RXC009 has shown a favourable absorption, distribution, metabolism and excretion (ADME) and safety profile with a Drug-Drug Interaction (DDI) assessment completed confirming its suitability for potential use in combination.
Discoidin domain receptors (DDRs) were discovered in the 1990s but more recently they have gained traction as new therapeutic targets with potential to treat multiple fibrotic conditions.
DDRs are receptor tyrosine kinases containing a discoidin homology domain in their extracellular region, which bind to collagen. There are two DDR receptors, DDR1 and DDR2, which act as non-integrin collagen receptors.
On binding of collagen, the DDRs autophosphorylate, initiating various downstream signalling pathways that drive clustering, upregulation and further collagen synthesis. DDR expression is increased in many fibrotic diseases and preclinical proof-of-concept for small molecule inhibitors has been demonstrated in preclinical models of lung and kidney fibrosis.
We have developed novel, orally available, selective and potent small molecule DDR inhibitors with drug-like properties, and in October 2023, nominated our DDR1 selective inhibitor as development candidate RXC009, which has the potential to be a first-in-class treatment option for chronic kidney disease.
Currently, there are no selective DDR1 inhibitors in clinical trials, although several kinase inhibitors developed for other kinases have shown varying degree of DDR specificity. We presented a poster on a novel, selective DDR1 inhibitor, REDX12271, at the American Society of Nephrology (ASN) Kidney Week, November 2022, and we returned to ASN in November 2023 to present RXC009 preclinical data from a therapeutic Unilateral Ureteral Obstruction (UUO) murine model of kidney fibrosis.
The data presented showed that RXC009 treatment resulted in:
- A significant reduction in histological markers of both inflammation, kidney injury and fibrosis.
- Target engagement in kidney tissue was also demonstrated in phospho-DDR1 (p-DDR1)
RXC009 has shown a favourable absorption, distribution, metabolism and excretion (ADME) and safety profile with a Drug-Drug Interaction (DDI) assessment completed confirming its suitability for potential use in combination.
Discoidin Domain Receptors (DDR) are novel, druggable therapeutic targets for fibrosis
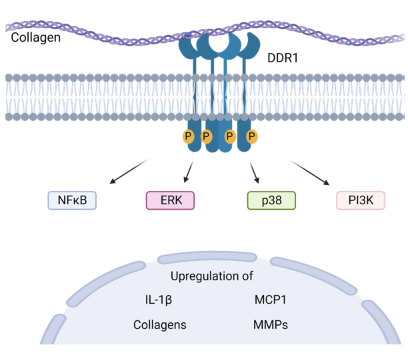
Figure 1: Discoidin Domain Receptors (DDRs) are collagen-activated receptor tyrosine kinases which have been shown to be highly expressed in many fibrotic diseases. On binding of collagen, the DDRs autophosphorylate, initiating various downstream signalling pathways that drive clustering, upregulation and further collagen synthesis. (There are two receptors of DDR: DDR1 and DDR2)
RXC009 Significantly Reduces Inflammation and Kidney Injury Associated With Chronic Kidney Disease in a Therapeutic Murine UUO Model
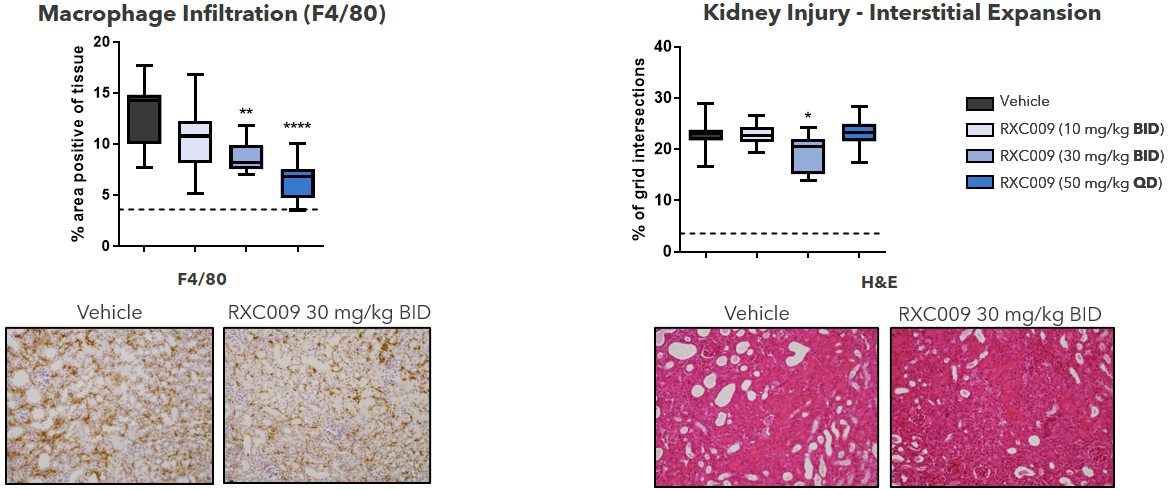
Figure 3: RXC009 in a 10-day therapeutic murine UUO kidney fibrosis model. Female C57BL/6J mice. Oral dosing from day 5-10 post-surgery. Terminal sampling analysis at T = 2 h. Inflammation (F4/80) and tubulointerstitial damage (H&E) as determined by immunohistochemistry.
Statistics: One-way ANOVA with Dunnett’s multiple comparison test calculated relative to vehicle control.
RXC009 Significantly Decreases Fibrosis, Collagen Deposition and Myofibroblast Transformation in a Therapeutic Murine UUO Model
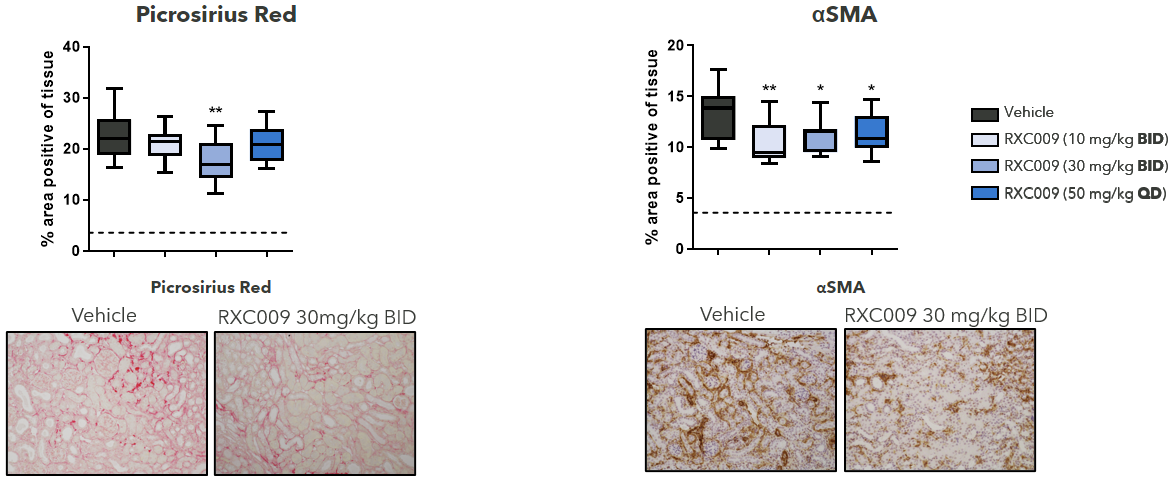
Figure 4: RXC009 in a 10-day therapeutic murine UUO kidney fibrosis model. Female C57BL/6J mice. Oral dosing from day 5-10 post-surgery. Terminal sampling analysis at T = 2 h. Fibrosis and collagen deposition (picrosirius red), myofibroblast transformation (α-SMA) as determined by immunohistochemistry.
Statistics: One-way ANOVA with Dunnett’s multiple comparison test calculated relative to vehicle control.